Centre of Thermodynamics of Processes






The CTP has 5 laboratories, among them a high safety laboratory

Especially developed for pressurized fluid sampling and sample analysis by gas chromatography
The Centre of Thermodynamics of Processes (CTP) is a joint research centre of MINES ParisTech and ARMINES and one of the 18 research centres of MINES ParisTech.
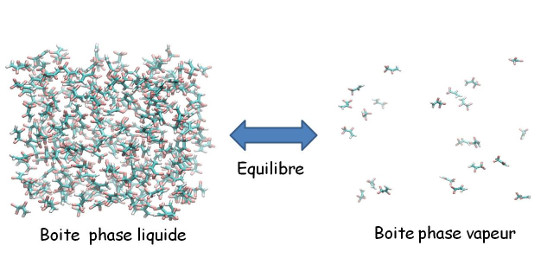
The thermodynamic and kinetic models are at the heart of all developments and optimization of industrial processes. Thus, the research topics of the CTP mainly concern fluid thermodynamics and the study of phase diagrams. The kinetic aspects are also dealt with since estimating transfer coefficients requires knowledge of thermo-physical properties such as density, heat capacities, viscosity, surface tension and thermal conductivity. Phase diagrams help to define the best process and to show how one can articulate the unit operations according to the temperature, pressure and composition conditions. The study of the phase diagrams also helps to understand the physicochemical phenomena in order to improve the functioning of the separation units: knowledge of the molecular aspects helps develop tomorrow's processes.
The heart of the work done at CTP is the thermodynamics of phases equilibria that combine the modelling work, the experimental work but also the design of innovative tools for the thermophysical properties measurement. The CTP develops and/or modifies existing experimental tools in order to carry out its work. It develops new experimental protocols including the calibration and the estimation of the measurement uncertainties. The CTP attaches great importance to data treatment and to the comparison with data from the literature.
To conclude, the CTP develops original thermodynamic and kinetic models in order to control the phenomena and predict the behaviours. The aspects related to the transfer of the software and/or the models to the industry are part of the concerns of the CTP's researchers.
The activities of the CTP are mainly part of Axes 2 and 3 of the Energy and Processes department, Decarbonation of the processes and New and Renewable Energies, but they can be extended to other fields since the fluid thermodynamics is a "pillar theme" of process engineering.
For more information about the general presentation of the CTP, click here.
CTP is also active in the fields of education and training. For more information, click here.

Ecole
240 ans de recherche et de formation
Vidéo : 240ans de recherche…
> En savoir +

Formation
Mines Paris plébiscitée par ses étudiantes
Mines Paris - PSL, une école qui répond…
> En savoir +

Formation
Femmes de science
Chercheuses confirmées, doctorantes, élèves ou alumni,…
> En savoir +

Formation
Quelle école d’ingénieurs a le…
Mines Paris - PSL au Top 5 du classement LinkedIn 2023…
> En savoir +

Formation
En l'honneur de Cécile DeWitt-Morette
Claude Ribbe (Mairie du 6e arrondissement), Armand Hatchuel…
> En savoir +

Formation
Classement "Employabilité" THE 2022
Dans l'édition 2022 du Global Employability…
> En savoir +
